Cancer immunotherapy, harnessing the body’s immune system to fight malignant cells, represents a groundbreaking shift in the oncology landscape and has marked a turning point in cancer treatment achieving remarkable results in patients. In recent years, immunotherapy has emerged as the therapeutic option of choice for several advanced cancers. However, accurate assessment of treatment response in immunotherapy is crucial. Unlike traditional chemotherapy, immunotherapy may not exhibit immediate tumour shrinkage, rendering conventional response criteria inadequate. The integration of Radiomics and Artificial Intelligence (AI) has the potential to revolutionise the field of immunotherapy response assessment, offering a promising avenue to enhance cancer treatment strategies.
Radiomics is a quantitative imaging process extracting and analysing high quantities of digital information from standard-of-care imaging. This new wealth of data opens the door to increased analytics and the possibility of building artificial intelligence-based models to assess clinical outcomes, including prognostic or prediction of response to treatment (1). Radiomic models can also be used to characterise the genotype of tumours, overall survival, the response of a disease to a given treatment or to assess the response to a treatment beyond the classic radiologic criteria (2).
As the effectiveness of immunotherapy varies among patients and especially among lesions, making precise local response assessments is crucial. With its ability to analyse vast datasets swiftly and accurately, AI could play a pivotal role in this endeavour. This blog post delves into the possible role of radiomics and advanced imaging analysis in evaluating immunotherapy responses, highlighting their importance in guiding clinical decisions and optimising patient outcomes.
Imaging plays a fundamental role in modern cancer management. It provides a non-invasive method to visualise tumours, assess their size, and monitor changes over time. Traditional modalities such as CT, MRI, and PET have been invaluable in tracking cancer progression (3). Furthermore, the emergence of specialised imaging techniques like Immuno-PET have opened new frontiers in assessing immunotherapy responses (4). These technologies offer insights into tumour biology, immune infiltration, and treatment-induced changes, facilitating a more comprehensive evaluation of therapeutic efficacy. Immunotherapy introduces unique complexities in response assessment. Pseudo progression, where tumours initially appear to grow before responding to treatment, can complicate interpretation (5). Conversely, in hyper progression an unexpected rapid progression in disease following immunotherapy can be registered (6). However, a significant challenge is posed by the heterogeneity within and between tumours, as some lesions may respond while others progress (7). Determining the optimal timing and frequency of assessments remains an ongoing issue. Addressing these challenges is vital for accurate response evaluation, ensuring that patients receive timely adjustments to their treatment plans.
Many drugs have received regulatory clearance both in the EU and US for melanoma, lung, bladder, renal and oropharyngeal cancers among others (8). In turn, the tools to assess therapy efficacy and response have evolved to better understand the disease progression and the effects of these new therapies. This is the case for example with RECIST criteria, originally developed for chemotherapy, was updated with immune-related response criteria (irRC), immune-related RECIST (irRECIST), and immune RECIST (iRECIST) (3). Image assessment of cancerous lesions development is still the state of the art for therapy planning and follow-up, but novel tools are required to improve patient management and quality of life.
The integration of radiomics and AI has the potential to revolutionise the field of immunotherapy response assessment, offering a promising avenue to enhance cancer treatment strategies. Immunotherapy effectiveness varies among patients and among lesions, making precise response assessment crucial. With its ability to analyse vast datasets swiftly and accurately, AI could play a pivotal role in this endeavour. AI algorithms can process a multitude of quantitative features extracted from radiological images, collectively known as radiomics features (9). These features encompass information about tumour heterogeneity, shape, and texture, which may hold the key to predicting immunotherapy response as well tumour biology.
Through machine learning models, AI can correlate radiomics features with clinical outcomes, helping clinicians stratify patients into different response categories or to predict the insurgence of immune-related adverse events. Moreover, radiomics can expedite the assessment process, enabling timely adjustments to treatment plans and reducing the burden of unnecessary side effects for non-responsive patients. This in turn could also improve the cost-effectiveness of the drug development life cycle, reducing the rate of failed clinical trials in oncology and costs for pharmaceutical companies.
Radiomics in practice:
Correlation of radiomic features with tumour biology
The field of radiomics offers additional information for the prediction of prognosis in patients treated with immunotherapy based on established molecular biomarkers. For example, Chen et al. (10) assessed the ability of radiomic features to predict tumour-infiltrating lymphocytes (TIL) status of a tumour in patients with hepatocellular carcinoma (HCC). Features extracted from MRI tumoral and peri-tumoral region were used to predict the immunoscore of patients, which assessed the density of CD3+ and CD8+ T cells within the tumour core and invasive margin. This study found that a radiomics model which incorporated both intra and peri-tumoral radiomic features had a higher AUC in the validation cohort compared to a model with tumoral features alone (0.89 vs. 0.63).
Several studies have already explored the association of radiomic features with PD-L1 status (11),(12),(13). One of the first studies assessing the ability of radiomic features to predict PD-L1 status was conducted by Jiang et al.(14) This was a retrospective single-centre study that extracted radiomic features from both the CT and PET scans of a pre-operative PET/CT in patients with lung cancer undergoing surgery. The PD-L1 status of patients was assessed on postoperative samples via immunohistochemistry testing. The model performed very well for both PD-L1 ≥ 1% and PD-L1 ≥ 50%. A significant strength of this study is the use of post-operative samples to detect PD-L1 status. Another study considering patients with Stage IV NSCLC, found a radiomics-based model predictive of PD-L1 ≥ 50% on biopsy samples with moderate performance (AUC 0.65) (15).
Other tissue biomarkers of interest include microsatellite instability (MSI) and tumour mutational burden (TMB) (13), (16). One study was able to validate a radiomics model incorporating 40 radiomic features to predict microsatellite instability in colon cancer with an AUC of 0.76 (17). Another study in patients with NSCLC developed both a CT-based radiomics and a deep learning model to predict TMB (AUC 0.74 and AUC 0.85, respectively) (18).
Prediction of immune-related adverse events
Several studies investigated the ability of radiomics to predict immune-related adverse events. Mu et al. were able to train and validate a radiomics model derived from PET/CT features of the primary tumour in patients with NSCLC undergoing ICI treatment to predict the risk of immune-related severe adverse events (19). The model also incorporated clinical features including type and dose of immunotherapy. Another study, analysing a mixed cohort of patients, developed a radiomics model based on CT-derived features from the lung to predict the risk of pneumonitis with 100% accuracy, however the number of patients in the study was quite small, with only 2 out of 32 patients experiencing pneumonitis (20). Some research has also been carried out on radiomics-based distinction between radiation and immune-induced pneumonitis (21), (22). This is a very promising avenue of research, tackling a highly relevant unmet clinical need.
Predicting response to immunotherapy
Individual patient response to specific therapy is a fundamental aspect of radiomics potential. Radiomics.bio conducted a retrospective study to determine the role of CT-based radiomics and delta-radiomics in predicting tumoral response and survival in patients with advanced NSCLC treated with PD-1/PD-L1 inhibitors (1).
Delta radiomics extracts radiomic features from the same region of interest of the same patient at different time intervals to study the variation of radiomic features over time. Studying the phenotypical modifications of tissues and or lesions that may occur following the introduction of treatment before any size modification bears a significant promise in correctly assessing immunotherapy, wherein the kinetics of tumour size is not always correlated to treatment response.
Our results, illustrated in Figure 1.0 below, demonstrated the ability of a CT-based delta-radiomics signature to perform early identification of patients who are more likely to benefit from PD-1/PD-L1 inhibitors. Thus, further confirming the ability of radiomics to provide a solution for the challenges encountered in the development of immunotherapies. Radiomics.bio’s delta-radiomics signature was able to identify patients who presented a clinical benefit at 6 months, with an AUC obtained on an external test dataset of 0.8 (95% CI: 0.65-0.95)(1).
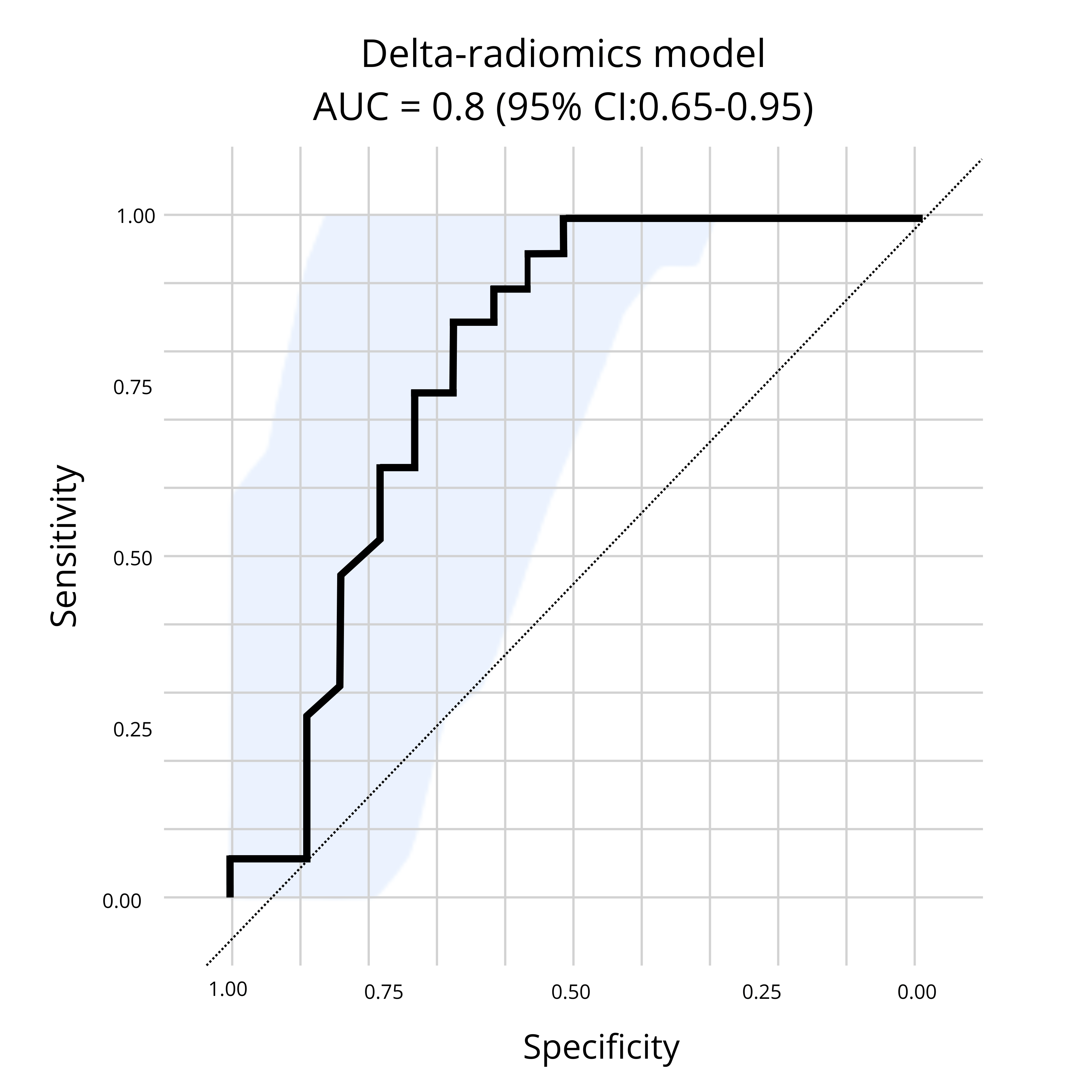
Figure 1: Presented at SITC,Radiomics.bio’s delta-radiomics signature demonstrates the ability to identify patients who presented a clinical benefit at 6 months, with an AUC obtained on an external test dataset of 0.8 (95% CI: 0.65-0.95), from baseline + first follow up CT scan.
Conclusion
Radiomics complements other modes of analysis by filling in gaps of knowledge through additional insights, moreover, the radiomic solution proposes multiple advantages including being non-invasive, repeatable over time and being able to explore the entirety of the primary tumour as well as all secondary lesions included in medical scans (1). By identifying quantitative features that are not identifiable by simple visual inspection, radiomics creates enhanced data models to allow for insight-based decision making (2).
About Radiomics
Radiomics is an imaging research organisation which focuses on AI powered healthcare, with a unique expertise in Radiomics, Deep Learning & Federated Learning applied to oncology and other therapeutic areas. Founded in 2016, Radiomics leverages its proprietary technology to support pharma and biotech companies in all phases of clinical development to extract the maximum information from clinical trial images. Based in Liège, Belgium, Radiomics offers advanced image analysis services to support both prospective and retrospective clinical trials to better understand observed treatment effect.
With collaborations in place with several hospitals and pharma companies, Radiomics is dedicated to its mission to enable the right diagnosis & treatment for the right patient through quantitative imaging and machine learning.
References
1. Cousin F, Louis T, Dheur S, Aboubakar F, Ghaye B, Occhipinti M, et al. Radiomics and Delta-Radiomics Signatures to Predict Response and Survival in Patients with Non-Small-Cell Lung Cancer Treated with Immune Checkpoint Inhibitors. Cancers. 2023 Jan;15(7):1968.
2. Porcu M, Solinas C, Mannelli L, Micheletti G, Lambertini M, Willard-Gallo K, et al. Radiomics and “radi-…omics” in cancer immunotherapy: a guide for clinicians. Crit Rev Oncol Hematol. 2020 Oct 1;154:103068.
3. Berz AM, Dromain C, Vietti-Violi N, Boughdad S, Duran R. Tumor response assessment on imaging following immunotherapy. Front Oncol. 2022;12:982983.
4. Dercle L, Sun S, Seban RD, Mekki A, Sun R, Tselikas L, et al. Emerging and Evolving Concepts in Cancer Immunotherapy Imaging. Radiology. 2023 Jan;306(1):32–46.
5. Frelaut M, du Rusquec P, de Moura A, Le Tourneau C, Borcoman E. Pseudoprogression and Hyperprogression as New Forms of Response to Immunotherapy. BioDrugs. 2020 Aug 1;34(4):463–76.
6. Sehgal K. Hyperprogression in Patients With Cancer Receiving Immune Checkpoint Inhibitors. JAMA Netw Open. 2021 Mar 1;4(3):e211839.
7. Pseudoprogression and hyperprogression after checkpoint blockade – ScienceDirect [Internet]. [cited 2024 Feb 22]. Available from: https://www.sciencedirect.com/science/article/abs/pii/S1567576918301218
8. Diseases | Free Full-Text | Emerging Trends in Immunotherapy for Cancer [Internet]. [cited 2024 Feb 22]. Available from: https://www.mdpi.com/2079-9721/10/3/60
9. Guiot J, Vaidyanathan A, Deprez L, Zerka F, Danthine D, Frix AN, et al. A review in radiomics: Making personalized medicine a reality via routine imaging. Med Res Rev. 2022;42(1):426–40.
10. Chen S, Feng S, Wei J, Liu F, Li B, Li X, et al. Pretreatment prediction of immunoscore in hepatocellular cancer: a radiomics-based clinical model based on Gd-EOB-DTPA-enhanced MRI imaging. Eur Radiol. 2019 Aug 1;29(8):4177–87.
11. Frontiers | Radiomics Study for Predicting the Expression of PD-L1 and Tumor Mutation Burden in Non-Small Cell Lung Cancer Based on CT Images and Clinicopathological Features [Internet]. [cited 2024 Feb 22]. Available from: https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2021.620246/full
12. Biomedicines | Free Full-Text | Multi-Omics Approaches for the Prediction of Clinical Endpoints after Immunotherapy in Non-Small Cell Lung Cancer: A Comprehensive Review [Internet]. [cited 2024 Feb 22]. Available from: https://www.mdpi.com/2227-9059/10/6/1237
13. Cancers | Free Full-Text | NSCLC Biomarkers to Predict Response to Immunotherapy with Checkpoint Inhibitors (ICI): From the Cells to In Vivo Images [Internet]. [cited 2024 Feb 22]. Available from: https://www.mdpi.com/2072-6694/13/18/4543
14. Assessing PD-L1 Expression Level by Radiomic Features From PET/CT in Nonsmall Cell Lung Cancer Patients: An Initial Result – ScienceDirect [Internet]. [cited 2024 Feb 22]. Available from: https://www.sciencedirect.com/science/article/abs/pii/S1076633219302338
15. Tian P, He B, Mu W, Liu K, Liu L, Zeng H, et al. Assessing PD-L1 expression in non-small cell lung cancer and predicting responses to immune checkpoint inhibitors using deep learning on computed tomography images. Theranostics. 2021 Jan 1;11(5):2098–107.
16. Lin ZF, Qin LX, Chen JH. Biomarkers for response to immunotherapy in hepatobiliary malignancies. Hepatobiliary Pancreat Dis Int. 2022 Oct 1;21(5):413–9.
17. Golia Pernicka JS, Gagniere J, Chakraborty J, Yamashita R, Nardo L, Creasy JM, et al. Radiomics-based prediction of microsatellite instability in colorectal cancer at initial computed tomography evaluation. Abdom Radiol N Y. 2019 Nov;44(11):3755–63.
18. He B, Dong D, She Y, Zhou C, Fang M, Zhu Y, et al. Predicting response to immunotherapy in advanced non-small-cell lung cancer using tumor mutational burden radiomic biomarker. J Immunother Cancer. 2020 Jul 6;8(2):e000550.
19. Mu W, Tunali I, Qi J, Schabath MB, Gillies RJ. Radiomics of 18F Fluorodeoxyglucose PET/CT Images Predicts Severe Immune-related Adverse Events in Patients with NSCLC. Radiol Artif Intell. 2020 Jan;2(1):e190063.
20. Colen RR, Fujii T, Bilen MA, Kotrotsou A, Abrol S, Hess KR, et al. Radiomics to predict immunotherapy-induced pneumonitis: proof of concept. Invest New Drugs. 2018 Aug 1;36(4):601–7.
21. Differentiation between immune checkpoint inhibitor‐related and radiation pneumonitis in lung cancer by CT radiomics and machine learning – Cheng – 2022 – Medical Physics – Wiley Online Library [Internet]. [cited 2024 Feb 22]. Available from: https://aapm.onlinelibrary.wiley.com/doi/full/10.1002/mp.15451
22. Chen X, Sheikh K, Nakajima E, Lin CT, Lee J, Hu C, et al. Radiation Versus Immune Checkpoint Inhibitor Associated Pneumonitis: Distinct Radiologic Morphologies. The Oncologist. 2021 Oct 1;26(10):e1822–32.
A new grant will support digital health solutions to advance precision medicine in cancer care, translating into personalized treatment matching and optimized outcomes for thousands of patients.
Montreal, September 5, 2023 – A pilot project led by the Jewish General Hospital proposes a new promising approach to cancer treatment known as data-based precision oncology to accelerate access to new and highly effective treatments for cancer patients. This innovative strategy involves tailoring cancer treatments based on intricate details unique to each patient’s tumor.
In the 1990s, the inception of precision oncology saw doctors identifying specific DNA mutations within tumors in an attempt to identify drugs capable of targeting these alterations.
The team aims to take precision medicine to new heights by not only analyzing the DNA of tumors but also incorporating medical images and microscopic characteristics of these tumors. Researchers will use this information to provide a more complete picture of the tumor, enhancing the ability to align the treatment more effectively with the patient’s specific tumor characteristics. Supported and accompanied by MEDTEQ+ through its IMPACT program, this ambitious undertaking involves a collaboration between esteemed entities: the McGill Centre for Translational Research in Cancer (MCTRC) from the Segal Cancer Centre (SCC) of the Lady Davis Research Institute (LDI) at the Jewish General Hospital (JGH), OROT – CIUSSS West-Central Montreal’s connected health innovation hub, Exactis Innovation – the National Centre of Excellence in Personalized Medicine. The collaboration of this project reaches beyond Quebec, with the inclusion of industry partner Radiomics, a Belgian (Liege) based biotech with a Quebec subsidiary since 2022 working within the domain of bespoke image analysis for oncology clinical trials and development of software as a medical device (ISO 13485 compliant). The project is also supported by the JGH Foundation and the Institute TransMedTech.
“The project emerged from recognizing the value of integrating multiple data in advancing precision medicine and cancer research. We plan to use computer tools and artificial intelligence to gather data in an automated fashion instead of doing it all manually. Right now, there are many big sets of data that no one is looking at simply because we cannot easily access this information,” says Professor Shirin Abbasinejad Enger, Tier II Canada Research Chair in Medical Physics, tenured Associate Professor at the Gerald Bronfman Department of Oncology, Director, Medical Physics Unit and Researcher at the MCTRC of the Lady Davis Research Institute of the Jewish General Hospital.
Pr. Abbasinejad Enger co-leads the project with Dr. Mark Basik, Principal Investigator of the PMT registry, Surgical Scientist at the LDI, and Head of the Cancer Genomics and Translational Research Laboratory at the SCC at the JGH: “By creating computer programs that use artificial intelligence and can analyze images and written information, we can get important data about each patient from their medical pictures, reports, and records and bring it together with the analysis of their tumors. These programs will help us determine the best treatment for each person.”
“Our project will enhance the quality and value of our already ongoing Personalize My Treatment (PMT) patient registry in partnership with the National Centre of Excellence in Personalized Medicine in Cancer – Exactis Innovation,” outlines Exactis Innovation CEO, Kostas Trakas. “As a pilot at the JGH, it will include four datasets: medical images, pathology, molecular and clinical data. This will enable cancer research to use more defined patient subpopulations for specific drug development, treatment selection and monitoring, and potentially prevention. We will then leverage the power of advanced image analysis and statistical modelling to gain insights into the database.”
The project is expected to have a wide-reaching impact:
- Cancer patients will benefit from a better treatment selection. Once the prediction algorithm is developed, it would be possible to identify that a traditional first-choice treatment would not be the optimal treatment for a specific patient based on analysis of the combined information on their pathology slides, genetic markers, and medical imaging. This can potentially shorten the patient’s time on treatment, reduce exposure to unnecessary cytotoxic treatments, and improve treatment outcomes.
- Cancer researchers will be empowered to develop new models for their research based on these data.
- Clinicians will have at their disposal models based on real-world data collected from their patients.
- Having access to such real-world data may help reshape Quebec and Canada’s drug approval and reimbursement programs by supporting avant-garde approaches that accelerate patient access to new and highly effective treatments.
- For the industry, it means the availability of unbiased multimodal data sets, which facilitates finding patient populations for clinical trials of new drugs.
Also, the project will generate long-term employment among the involved partners, with the hope of extending the Exactis Innovation network to other sites in Quebec hospitals, and, subsequently to other provinces.
The collaborative research project, which is entitled “Digital Health to Advance and Support Precision Oncology”, is supported by a grant of $358 825 from the Ministry of Economy, Innovation and Energy (MEIE) within the framework of the MEDTEQ+ IMPACT program and by $75,000 from TransMedTech Institute. This amount was exceeded by the contributions of other public and private funding partners, the JGH Foundation, Exactis Innovation, Mitacs and Radiomics.
“As part of its mission, MEDTEQ+ is proud to support and contribute to this innovative and collaborative research project. We are delighted to financially support this digital health solution to advance precision medicine in cancer care through our IMPACT program, funded by the Ministère de l’Économie, de l’Innovation et de l’Énergie du Québec (MEI),” said Iris Leroux, vice-president, Collaboration and Operations at MEDTEQ+ Consortium. “In addition to meeting all the objectives of our funding program – such as facilitating knowledge transfer, accelerating the integration of new technologies and care pathways within the health system, and supporting the creation of new markets for consortium partners – it offers potential solutions to improve patient care and addresses healthcare system challenges.”
“The TransMedTech Institute is privileged to be associated with and contribute to this visionary, innovative and collaborative initiative, which addresses important issues in our healthcare system,” says Pr. Carl Eric Aubin, Executive and Scientific Director. “This project is fully in line with TransMedTech’s vision of making innovation in medical technologies an asset for population health and socio-economic development. As part of our mission, we are committed to supporting this innovative initiative, in order to facilitate and catalyze its development and implementation.”
The project’s findings and advancements could have lasting effects on precision medicine, shaping the future of cancer treatment and management.
Project Partners
For further information or to arrange interviews with Professor Shirin Abbasinejad Enger or Dr. Mark Basik, please contact:
Pascal Fischer
Research Communications Officer | Lady Davis Institute
Tel.: 514 340-8222 x 28661 or 450 218-6487
For more information about the Lady Davis Institute: www.ladydavis.ca
For more information about the Jewish General Hospital: www.jgh.ca
For more information about the Jewish General Hospital Foundation: www.jghfoundation.org
For more information about CIUSSS West-Central Montreal: https://www.ciussswestcentral.ca/
For more information about the MCTRC: www.mcgill.ca/translational-research-cancer/
For more information about OROT: https://orot-jgh.org/
For more information about MEDTEQ+: www.medteq.ca
For more information about TransMedTech Institute: www.transmedtech.org
For more information about Exactis Innovation: www.exactis.ca
For more information about Radiomics: www.radiomics.bio
About Radiomics
Radiomics is an imaging research organisation which focuses on AI powered healthcare, with a unique expertise in Radiomics, Deep Learning & Federated Learning applied to oncology and other therapeutic areas. Founded in 2016, Radiomics leverages its proprietary technology to support pharma and biotech companies in all phases of clinical development to extract the maximum information from clinical trial images. Based in Liège, Belgium, Radiomics offers advanced image analysis services to support both prospective and retrospective clinical trials to better understand observed treatment effect.
With collaborations in place with several hospitals and pharma companies, Radiomics is dedicated to its mission to enable the right diagnosis & treatment for the right patient through quantitative imaging and machine learning.
DeuterOncology joins forces with Radiomics to develop a clinical tool to predict patient response to treatment.
DeuterOncology obtains SPW grant funding
Liège, Belgium, April 07, 2023 – DeuterOncology, a Liège-Based (BE) biotech company, is pleased to announce it has successfully been awarded the Win4Company grant by the Wallonie recherche SPW. The clinical-stage drug development company has been granted 478,000 EUR for establishing a partnership with a Liège-based biotech, Radiomics, for the development and validatation of an AI-based imaging biomarker as a potential companion diagnostic for a novel best-in-class deuterated MET kinase inhibitor (DO-2) as an optimized personalized cancer therapy.
The grant brings together the expertise of two Belgian biotech companies to develop AI-based CT imaging for identifying subsets of lung cancer patients that have MET gene alterations. The grant will also enable DeuterOncology to improve the determination of clinical response to DO-2 by the accurate determination of tumour volume change.
“We are excited to receive this grant from the SPW, which will enable us to collaborate with Radiomics and develop cutting-edge AI-based CT imaging technology”- said Dr. Timothy Perera, CEO of DeuterOncology. This breakthrough technology will provide oncologists with an easier way of identifying patients with MET oncogene driven tumours using standard imaging and reduce the need for painful biopsies and timeconsuming testing to identify potential responders.
“We are very pleased to establish this local collaboration with DeuterOncology and to be part of such a ground-breaking trial which could have global implications. Optimizing decision making is fundamental to improving patient treatment, allowing medical professionals to make informed decisions will drastically improve drug development trajectories” – Dr Carlos Meca, CCO of Radiomics.This collaboration will combine DeuterOncology’s expertise in cancer research and genomics with Radiomics’ expertise in advanced AI-based image analysis technology to develop a powerful tool for predicting patient response to treatment.
This grant is an important milestone for DeuterOncology and demonstrates the company’s commitment to developing targeted therapy that will improve patient outcomes. The joint project will help expand the strong drug development eco-system that is being created in the Walloon region.
About DeuterOncology
DeuterOncology is a clinical-stage biotech company focusing on the development of a first-in-class dual MET and RAS pathway inhibitor for targeted cancer therapies. The company has generated preclinical validation of its lead candidate DO-2, a novel, highly selective, brain penetrant deuterated MET and RAS pathway inhibitor that was licensed (worldwide rights outside of greater China) from OCTIMET Oncology NV and originally licensed in 2017 from Janssen Pharmaceutica. DeuterOncology has initiated a phase I clinical study to validate DO-2 as a best-in-class MET kinase inhibitor as suggested by the strong preclinical data package.
Based in Liège, the company was founded in September 2020 by Dr. Timothy Perera. DeuterOncology works together with an established broad network of collaborators across the world to achieve its mission of bringing new solutions to cancer patients.
For more information about DeuterOncology’s research, please visit our website at www.deuteroncology.com.
For more information on the ongoing clinical trial with DO-2, please visit ClinicalTrials.gov
About Radiomics
Radiomics is an imaging research organisation which focuses on AI powered healthcare, with a unique expertise in Radiomics, Deep Learning & Federated Learning applied to oncology and other therapeutic areas.
Founded in 2016, Radiomics leverages its proprietary technology to support pharma and biotech companies in all phases of clinical development to extract the maximum information from clinical trial images. Based in Liège, Belgium, Radiomics offers advanced image analysis services to support both prospective and retrospective clinical trials to better understand observed treatment effect.
With collaborations in place with several hospitals and pharma companies, Radiomics is dedicated to its mission to enable the right diagnosis & treatment for the right patient through quantitative imaging and machine learning.
For more information about Radiomics’ research, please visit https://radiomics.bio
Contact
PERERA, Timothy
CEO
tperera@deuteroncology.com
+32473558353
Paving the way for better diagnosis, treatment and care in pulmonary fibrosis
Three Lakes Foundation brings together academia, clinicians and industry to identify imaging insights for early detection of interstitial lung disease/pulmonary fibrosis (ILD/PF).
Please fill in your details below and get access to all of the elements of our information package such as our latest poster on the “Delta-radiomics to predict response and survival in solid tumors after immune checkpoint inhibitors therapy.”
NORTHBROOK, Ill., November 10, 2022 — Three Lakes Foundation has announced the expansion of B4PF, an initiative focused on at-risk individuals for ILD/PF, early diagnosis and therapeutic development in collaboration with Weill Cornell Medicine, Vanderbilt University Medical Center (VUMC) and Radiomics, an imaging research organization based in Liege, Belgium.
Together these collaborators exemplify Three Lakes Foundation’s commitment to improving time to diagnosis, accelerating new therapies and enhancing quality of life for those affected by ILD/PF, leading to better disease management and treatment options.

“We aim to investigate which insights generated from standard, computed tomography (CT) images can lead to earlier identification of patients with progressive pulmonary fibrosis,” said Anna Podolanczuk, M.D., an assistant professor of medicine at Weill Cornell Medicine. Dr. Podolanczuk will work closely with Jonathan Kropski, M.D., an associate professor of medicine at VUMC, the leading clinical partners in this research.
Radiomics’ expertise will help bring together a multi-disciplinary consortium to conduct key research for the early identification and diagnosis of ILD/PF via imaging.
“This project brings together the forefront of imaging, statistical modeling, diagnostics and industry to better understand the early signs of ILD/PF from an imaging perspective” says Cheryl Nickerson-Nutter, Ph.D. Dr. Nickerson-Nutter is executive vice president of research and development at Three Lakes Foundation. “Our foundation is dedicated to working with collaborative teams focused on changing the course of diagnosis, management and treatment of patients diagnosed with ILD/PF. This study will allow imaging experts, physicians and other industry leaders to evaluate, understand and best apply imaging-based early diagnostics.”
The project will be guided by internationally recognized industry and clinical experts to be announced at a later date. It will explore the value of imaging insights indicating early signs of ILD or PF in relatives of patients who have already been diagnosed with PF and in individuals with incidentally identified abnormalities on chest CT.
With over five million patients across the world affected by PF and 50,000 patients diagnosed with IPF annually in the U.S., the main objective of Three Lakes Foundation is to finally put an end to misdiagnosis, lack of awareness and information, and limited treatment options, resulting in an improvement in the patient journey and patients’ quality of life and generation of effective treatment options.
For more information contact:
Info@radiomics.bio
About VUMC:
Founded in 1875, Vanderbilt University Medical Center is a patient care, medical institution, and research enterprise with the mission of advancing personalised healthcare. Managing more than 3 million patient visits each year, VUMC is a resource for patients and clinicians throughout Tennessee and beyond to provide advanced care for complex and rare conditions. To learn more, visit: vumc.org.
About Radiomics:
Radiomics is an imaging research organization which focuses on AI powered healthcare, with a unique expertise in Radiomics, Deep Learning & Federated Learning applied to oncology and other therapeutic areas. Founded in 2016, Radiomics has its HQ in LégiaPark in Liege, Belgium and a North American office in Toronto, Canada. To learn more, visit: radiomics.bio.
About Three Lakes Foundation:
Three Lakes Foundation is a private non-profit organization dedicated to serving as a catalyst for uniting research, industries and philanthropy in pulmonary fibrosis. Three Lakes Foundation connects entrepreneurs, advocates and institutions to an innovation ecosystem that will transform approach to improve time to diagnosis and accelerate new therapies. To learn more, visit threelakesfoundation.org.
After 4 years, Radiomics is moving to LégiaPark
Liege (BE), 10/14/2022 -On September 19th, Radiomics officially opened doors to their new office at LégiaPark to provide for their ever-growing team and clients with the support they need. In a brand new 800m2 space, Radiomics will continue to serve its clients, develop its internal R&D and provide a modern and pleasant work environment for its most important asset: its collaborators.
Additionally, Radiomics will host young partner companies in its internal incubator and is already looking into applications.

“The objective of LégiaPark is to provide Life Science companies with workspaces adapted to their needs, providing customised offices, laboratories and cleanrooms, fully in line with the socio-economic context of our time, creating a business park with high energy and environmental performance. Our ambition at LégiaPark is to encourage commercial development by enabling companies to work together on a daily basis.” – Aurore Duijsens – CEO of LégiaPark.
An obvious choice for Radiomics, LégiaPark allows the organisation to go above and beyond its own sustainability policy, with green roofs, biodiversity friendly features, rainwater tanks and much more. “We are very excited to be moving to this new hub in Liege’s vibrant life sciences ecosystem. Not only for the additional need for space, but also the proximity to other companies in the healthcare space and to the hospital of MontLégia, were key contributors to our choice for LégiaPark. As one of its first occupants we are looking forward to the future.”- Wim Vos, CEO of Radiomics.
About Radiomics
Radiomics is an imaging research organization which focuses on AI powered healthcare, with a unique expertise in Radiomics, Deep Learning & Federated Learning applied to oncology and other therapeutic areas. Founded in 2016, Radiomics has its HQ in LégiaPark in Liege, Belgium and a North American office in Toronto, Canada. For further information, visit www.radiomics.bio and connect through Linkedin or Twitter.
Media contacts
Soraly Nzanga,
Junior Marketing Officer
info@radiomics.bio
Radiomics has created a digital information package for ESMO Congress to reduce our carbon footprint.
Sign up to get access to our information package including our brochures, papers and more. Radiomics is commited to intergrating sustainability into all of its activities. In order to do so, an online version of our information package was created to minimise our impact on the environment.
Please fill in your details below and get access to all of the elements of our information package such as our latest brochure on Leveraging Radiomics in Early Phase Clinical Development.
Radiomics’ digital event package for Immuno-Oncology Xchange 2022
Sign up to get access to our brochures, papers and more. Radiomics is commited to intergrating sustainability into all of its activities. In order to do so an online version of our information package was created to reduce our carbon footprint.
Please fill in your details below and get access to all of the elements of our information package such as our latest poster on the “Delta-radiomics to predict response and survival in solid tumors after immune checkpoint inhibitors therapy.”
The 1st Edition of Radiomics’ Newsletter Series
A message from our CEO:
“I am very happy to see the first issue of our Radiomics’ newsletter go live. Radiomics is taking the next steps to further advancing its promise of optimising decision making and enhancing insights for medical treatments, leading to better diagnosis and precision medicine and allowing us all to progress in the treatment of oncology and other fields. Striving to make an impactful change, we are very excited to share this part of our journey with you all.” – Wim Vos
Radiomics is pleased to announce its Quality Management System has been certified to be fully compliant with EN ISO 13485:2016 Medical Devices – Quality Management Systems – Requirements for regulatory purposes) standard by the notified body IMQ.
The ISO 13485:2016 standard is based on the ISO 9001 and specifies the requirements an organization needs to demonstrate for the design, development, production, storage, distribution, installation or servicing of a medical device. The standard ensures, through the implementation of a quality management system, that all medical devices are safe and meet the applicable regulatory compliance laws and customer needs.
Steven Lauwers, Chief Operational Officer at Radiomics: “We strive to achieve the highest standards in everything that we do. Therefore we decided to set the bar for all our operations as high as possible, being compliant to ISO13485:2016. This certification is a collective achievement of all the employees of Radiomics spearheaded by our QA team. We believe this certification, and the others that will follow, show our continuous dedication to go the extra mile.”
This certificate covers the design and development of Software as a Medical Device used for the diagnosis, prognosis or prediction of disease progression which are typically assessed through routine clinical imaging. As Radiomics is doing more than just designing and developing medical devices, it has decided that Radiomics’ quality management system should also cover our other activities.
With this certification, we also wanted to send a very clear message to all our research collaborations and clients positioning Radiomics as a trustworthy and reliable partner for all collaborations.
About Radiomics
Founded in 2016 and based in Liège, Radiomics is a Belgian AI powered research organization for next-generation image analysis, based on the unique experience of its founders, pioneers in radiomic science. Oncology being radiomics initial focus area, we have developed promising applications in various other therapeutic areas, such the respiratory field. Radiomics uses its advanced image analysis technology based on AI, deep learning, machine learning, and federated learning to obtain quantitative biomarker measurements from medical imaging, in a repeatable, reliable, and relevant manner. Its goal is to support decision-making through insights and optimize pharmaceutical and biotech companies’ clinical trials and drug development studies and provide clinicians with a patient-centered approach based on personalized medicine.
Radiomics has created a digital information package for the American Society of Clinical Oncology (ASCO) to reduce our carbon footprint.
Sign up to get access to our information package including our brochures, papers and more. Radiomics is commited to intergrating sustainability into all of its activities. In order to do so, an online version of our information package was created to minimise our impact on the environment.
Please fill in your details below and get access to all of the elements of our information package such as our latest brochure on Leveraging Radiomics in Early Phase Clinical Development.